The photoactive nitrogen impurity in nitrogen-doped zirconium titanate (N-ZrTiO4): a combined electron paramagnetic resonance and density functional theory study†
Abstract
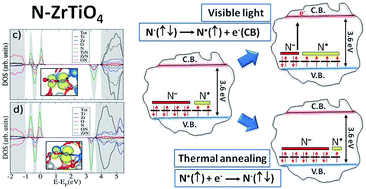
Nitrogen doping represents an important strategy to modulate the optical, magnetic and photochemical properties of oxides for applications spanning from photocatalysis to optoelectronics and spintronics. In this work for the first time zirconium titanate, a material that exhibits many attractive properties, including an excellent dielectric constant, high corrosion resistance, high permittivity at microwave frequencies and excellent temperature stability, has been doped with nitrogen via a wet chemistry method. A detailed description of the geometrical and the electronic structure of the dopant centre has been obtained coupling Electron Paramagnetic Resonance (EPR) spectroscopy and density functional theory (DFT) calculations. Insertion of nitrogen impurities into the ZrTiO4 lattice modifies the optical properties causing an appreciable absorption in the visible range. The joint analysis of the EPR evidence and the DFT elaboration indicates that the nitrogen impurities preferentially occupy an interstitial position of the lattice generating intra-band gap states about 1 eV above the valence band edge that are responsible for the visible light absorption. The majority of the intra-band gap states are diamagnetic (N−) while a minor fraction is paramagnetic (N˙). These centres are photosensitive in that the ratio between N− and N˙ is modified, upon irradiation, because of electron excitation from the intra-band gap states to the conduction band.


 Please wait while we load your content...
Please wait while we load your content...