A temperature-responsive poly(vinyl alcohol) gel for controlling fluidity of an inorganic phase change material†
Abstract
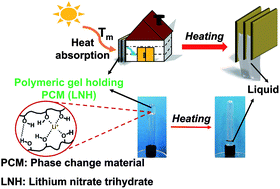
An environmentally responsive polymeric network has been demonstrated for incorporating an inorganic salt hydrate phase change material (PCM). The unique feature of this material is the capability of a polymer gel to simultaneously provide shape stabilization of a liquid salt hydrate phase at a lower temperature, and to reversibly adjust its assembly strength in response to a temperature increase. This approach enables convenient control over the viscosity of the PCM gel system, allowing for filling and removal of the mixture in a liquid state but operation in the solid/gel regime. The system consists of poly(vinyl alcohol) (PVA) dissolved in lithium nitrate trihydrate (LNH), a high-latent-heat salt hydrate PCM. Dynamic light scattering (DLS) and viscometry studies indicate that in dilute solution, PVA chains are more expanded in LNH than in water. At a concentration of 15 wt% PVA, the PVA/LNH system – unlike PVA aqueous solutions – gels and exhibits a temperature-triggered gel-to-sol transition. Gelation is achieved only with PVA with the highest degree of hydrolysis (98%), with the gelation temperature of 20 ± 1 °C as measured by DLS and rheology. ATR-FTIR spectroscopy shows that the PVA gelation is enhanced due to dehydration of PVA chains in LNH, as compared to PVA aqueous solutions. Physically crosslinked PVA/LNH gels demonstrate highly repeatable and reproducible performance during ten cycles of LNH melting/freezing, as shown by differential scanning calorimetry (DSC). Taken together, these results confirm that PVA-based gels are promising matrices for hosting inorganic PCMs that withstand repeated melting/freezing cycling, but can be easily filled or drained as needed by raising the temperature well above the gelation temperature.


 Please wait while we load your content...
Please wait while we load your content...