Grafting of nano-Ag particles on –SO3−-coated nanopolymers for promoting methane hydrate formation†
Abstract
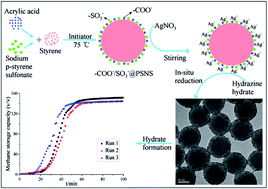
Sodium dodecyl sulfate (SDS) has been reported to be the most efficient promoter for hydrate-based natural gas storage and transportation, however, foam generation during hydrate dissociation seriously affects its application. Nano-metal particles have also been demonstrated to be efficient promoters, nevertheless their poor stability is a serious problem. In this work, we first fixed –SO3− groups (similar to the hydrophilic group of SDS) covalently on polystyrene through soap-free emulsion polymerization to synthesize –SO3−-coated nanopolymers, and by tuning their morphology they existed as amorphous polystyrene macromolecules (–SO3−@PSMM) or uniform polystyrene nanospheres (–SO3−@PSNS). Afterwards, we grafted nano-Ag particles with the size of 2–5 nm on the –SO3−-coated nanopolymers through electrostatic adsorption and in situ reduction to prepare Ag&–SO3−-coated nanopolymers (denoted as Ag&–SO3−@PSMM and Ag&–SO3−@PSNS), which were then used for the first time to promote methane hydrate formation. When 0.5 mmol L−1 amorphous Ag&–SO3−@PSMM was used at an initial pressure of 6 MPa and temperature of 275.15 K, the induction period was 32.2 ± 7.9–60.8 ± 14.2 min, the growth period was 108.8 ± 8.2–177.1 ± 38.9 min and the methane storage capacity reached 143.9 ± 3.7–145.2 ± 1.2 v/v, whereas when the spherical Ag&–SO3−@PSNS was used at 0.5 mmol L−1, the induction period and growth period were 17.8 ± 2.8–38.5 ± 8.0 and 39.6 ± 2.8–42.1 ± 0.9 min, respectively; and the storage capacity reached 149.3 ± 1.2–151.3 ± 3.0 v/v, indicating that Ag&–SO3−@PSNS were much better promoters compared with Ag&–SO3−@PSMM. Moreover, Ag&–SO3−@PSNS exhibited excellent recycling performance for 10 cycles of methane hydrate formation–dissociation. To sum up, the Ag&–SO3−-coated nano-promoters developed in this work showed significant potential in achieving the industrial application of hydrate-based natural gas storage and transportation.


 Please wait while we load your content...
Please wait while we load your content...