Surface structure and anion order of the oxynitride LaTiO2N†
Abstract
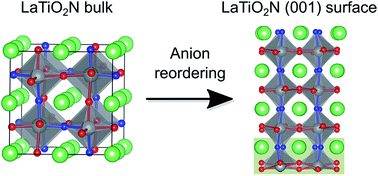
Oxynitrides are promising materials for water splitting under visible light. Members of this class of semiconductors that crystallise in the perovskite structure are often characterised by O/N disorder, while some studies observe 2D cis-chain ordering of the M–N–M bonds in the bulk. Despite the fact that the surface structure and composition is expected to have a significant influence on the surface chemistry and therefore the photocatalytic activity, little is known about the O/N arrangement at surfaces of these materials. In the present study, we investigate the surface structure of LaTiO2N, a particularly promising candidate for water splitting, using density functional theory (DFT) calculations. Based on slab calculations with different anion order we find that the N atoms prefer to form trans-chains at the (001) surface, as opposed to the bulk. This is governed by the electrostatic stability that is optimal for alternating charge-neutral (LaN)–(TiO2) atomic layers. We show that polar surfaces that do not fulfil this requirement will electronically or structurally reconstruct. Our results predict that in contact with vacuum, the LaTiO2N (001) surface will preferentially be LaN-terminated.


 Please wait while we load your content...
Please wait while we load your content...