Hydrazine-assisted formation of ultrathin MoS2 nanosheets for enhancing their co-catalytic activity in photocatalytic hydrogen evolution†
Abstract
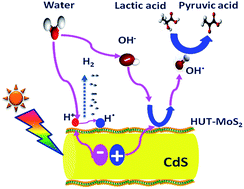
We demonstrate a simple and effective approach to modulate the active sites and electronic properties of MoS2 using hydrazine assisted liquid exfoliation for enhancing its co-catalytic activity in photocatalytic hydrogen evolution. The resulting ultrathin MoS2 (HUT-MoS2) nanosheets integrated on CdS nanorods, from hydrazine assisted liquid exfoliation, exhibit an excellent hydrogen evolution rate of 238 mmol g−1 h−1 under natural sunlight, which is the best performance ever reported for CdS/MoS2-based nanostructures. The apparent quantum yield reached 53.3% at 425 nm in 5 h. The hydrogen evolution rate was influenced by several experimental parameters, such as the photocatalyst dose, sacrificial donor concentration, and amount of co-catalyst on CdS, which were investigated in detail. More importantly, the CdS/HUT-MoS2 nanocomposite showed remarkable photo-stability for up to 100 h. The excellent hydrogen evolution performance and stability may be due to the unique structure and properties of HUT-MoS2 nanosheets, which significantly boosted the charge transportation between CdS and HUT-MoS2 and suppressed charge recombination, thus favoring the involvement of more electrons in hydrogen production. We believe that the presented nanohybrid design strategy and the implementation of this noble metal-free co-catalyst enable the development of inexpensive robust co-catalysts for sustainable hydrogen production to satisfy the growing global energy demand and address environmental problems.


 Please wait while we load your content...
Please wait while we load your content...