Tuning CH3NH3Pb(I1−xBrx)3 perovskite oxygen stability in thin films and solar cells†
Abstract
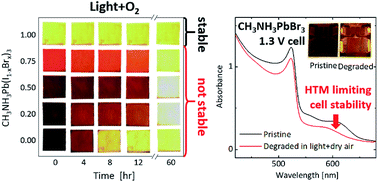
The rapid development of organic–inorganic lead halide perovskites has resulted in high efficiency photovoltaic devices. However the susceptibility of these devices to degradation under environmental stress has so far hindered commercial development, requiring for example expensive device encapsulation. Herein, we have investigated the stability of CH3NH3Pb(I1−xBrx)3 [x = 0–1] thin films and solar cells under controlled humidity, light, and oxygen conditions. We show that higher bromide ratios increase tolerance to moisture, with x = 1 thin films being stable to 120 h of moisture stress. Under light and dry air, partial bromide (x < 1) substitution does not enhance film stability significantly, with the corresponding solar cells degrading within two hours. In contrast, CH3NH3PbBr3 films show excellent stability, with device stability being limited by the organic interlayer. For these x = 1 films, we show that charge carriers are quenched in the presence of oxygen and form superoxide; however in contrast to perovskites containing iodide, this superoxide does not degrade the crystal. Our observations show that iodide limits the oxygen and light stability of CH3NH3Pb(I1−xBrx)3 perovskites, but that CH3NH3PbBr3 provides an opportunity to develop inherently stable high voltage photovoltaic devices and 4-terminal tandem solar cells.
- This article is part of the themed collection: International Year of the Periodic Table : From Pb and Sn Perovskites to the Next Generation


 Please wait while we load your content...
Please wait while we load your content...