Synthesis of nickel chalcogenide hollow spheres using an l-cysteine-assisted hydrothermal process for efficient supercapacitor electrodes†
Abstract
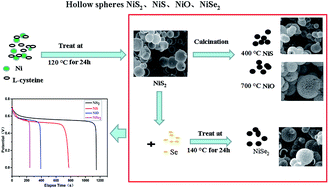
Transition metal materials, NiS, NiO and NiSe2, with a uniform hollow-structure were obtained based on NiS2 hollow spheres, and were successfully synthesized with an L-cysteine assisted hydrothermal method at 120 °C. The electrochemical properties of the NiS2, NiS, NiO and NiSe2 hollow sphere electrodes were investigated by cyclic voltammetry, galvanostatic charge–discharge and electrochemical impedance spectroscopy tests. Firstly, the cyclic voltammetry curves of the four materials all displayed pseudo-capacitance behaviour. Moreover, the galvanostatic charge–discharge tests showed that the specific capacitance of the NiS2, NiS, NiO and NiSe2 electrodes was 1643 F g−1, 1076 F g−1, 581 F g−1 and 341 F g−1, respectively, at a current density of 1 A g−1. After 1000 cycles, the specific capacitance of the NiS2, NiS, NiO and NiSe2 electrodes was retained at 455 F g−1, 368 F g−1, 246 F g−1 and 269 F g−1, respectively. The results implied that both the NiS2 and NiS electrodes exhibited higher specific capacitance, while the NiO and NiSe2 electrodes showed a longer cycling life. This work highlights the advantageous electrochemical performance of the different nickel compounds, and contributes to providing a good reference for the preparation of superior supercapacitor materials with high performance.


 Please wait while we load your content...
Please wait while we load your content...