Hemin-mediated construction of iridium oxide with superior stability for the oxygen evolution reaction†
Abstract
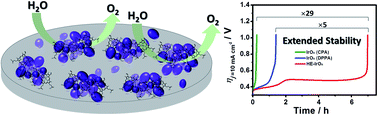
The biological molecule chloroprotoporphyrin IX iron(III) (hemin) was investigated as a novel catalyst to reduce the oxygen produced in IrOx-electroflocculation preparation and aid the catalytic stability of IrOx for the oxygen evolution reaction (OER) in both alkaline and acidic solutions. Hemin was deposited on glassy carbon (GC) electrodes through electropolymerisation with approximately 42% coverage and this mitigated the detrimental effect of accumulated oxygen on the subsequent growth of IrOx. When the IrOx catalyst was fabricated through 5 min of electroflocculation (coverage of approximately 8.43 × 10−8 mol cm−2 Ir), the stability time (the time required to reach the overpotential of 1.0 V at 10 mA cm−2) was approximately 7 h, which is 5-fold higher than that of the process without polymerised hemin, and 29.1-fold higher than that of the electroflocculation method reported in the literature. With the aid of hemin-catalysed oxygen depletion, the IrOx at the interface with the polymerised hemin is apparently deposited in a smaller and well-defined structure. The in situ spectroelectrochemical results revealed a lower dissolution rate for this specific IrOx structure, which facilitated subsequent electroflocculation towards the formation of a continuously structured deposit resistant to degradation. In 1 M NaOH, the IrOx catalyst sustained electrolysis (10 mA cm−2) for more than 20 h. In 1 M H2SO4, the overpotential (ηt=24 h) remained below 0.35 V. To the best of our knowledge, this is one of the few studies that have demonstrated a hydrous IrOx catalyst that can maintain its stability for over 24 h of the OER.


 Please wait while we load your content...
Please wait while we load your content...