Boron phosphide monolayer as a potential anode material for alkali metal-based batteries†
Abstract
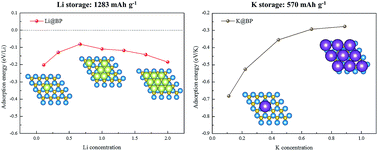
In this work, we adopt a first-principles study to evaluate the potential of boron phosphide (BP) monolayer as an anode material for alkali metal-based (e.g., Li, Na and K) batteries. It is found that the BP monolayer shows negative adsorption energies of −0.202, −0.160 and −0.681 eV, respectively, for Li, Na and K. During loading, when any of the three alkali metal atoms reaches a critical position (∼6 Å), the adsorption energy increases dramatically with no energy barrier. It is also shown that after adsorbing Li, Na or K, the semiconducting BP monolayer is transformed to a metallic state, becoming an electrical conductor. More importantly, the alkali metal atoms show high diffusivities on the BP monolayer, with low energy barriers of 0.364, 0.217 and 0.155 eV for Li, Na and K, respectively. Finally, the BP monolayer has high theoretical specific capacities of 1283 and 570 mA h g−1 for Li and K storages, which are among the highest values of the anode materials reported in the literature, and multiple times higher than that of graphite anode materials in use. The above-mentioned results suggest that the BP monolayer is a promising anode material not only for Li-ion and K-ion batteries, but also for other lithium-based and potassium-based batteries.


 Please wait while we load your content...
Please wait while we load your content...