Combinatorial screening of Pd-based quaternary electrocatalysts for oxygen reduction reaction in alkaline media†
Abstract
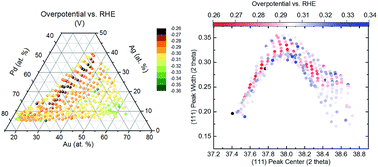
The implementation of electrochemical systems such as fuel cells has been hindered by the slow development of low cost high activity catalysts. Here we examine the oxygen reduction reaction performance of a combinatorial Pd–Au–Ag–Ti thin film library using high-throughput screening and correlate the electrochemical behavior to the crystallographic properties. We find compositions of ca. 40–60 at% Pd and 30–35 at% Au exhibit both a low overpotential of close to the value of pure Pt as well as high current density. We also observe a volcano-like relationship between the overpotential and the solid formation strain. This study provides compositional guidance towards the future synthesis of nanostructured quaternary Pd–Au–Ag–Ti alloys and suggests the potential for broader application of high-throughput electrochemical characterization by means of an automatic scanning droplet cell.


 Please wait while we load your content...
Please wait while we load your content...