Ergodicity breaking of iron displacement in heme proteins†
Abstract
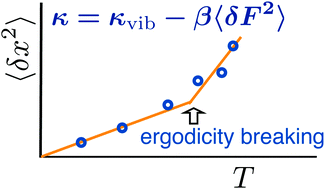
We present a model of the dynamical transition of atomic displacements in proteins. Increased mean-square displacement at higher temperatures is caused by the softening of the force constant for atomic/molecular displacements by electrostatic and van der Waals forces from the protein–water thermal bath. Displacement softening passes through a nonergodic dynamical transition when the relaxation time of the force–force correlation function enters, with increasing temperature, the instrumental observation window. Two crossover temperatures are identified. The lower crossover, presently connected to the glass transition, is related to the dynamical unfreezing of rotations of water molecules within nanodomains polarized by charged surface residues of the protein. The higher crossover temperature, usually assigned to the dynamical transition, marks the onset of water translations. All crossovers are ergodicity breaking transitions depending on the corresponding observation windows. Allowing stretched exponential relaxation of the protein–water thermal bath significantly improves the theory–experiment agreement when applied to solid protein samples studied by Mössbauer spectroscopy.


 Please wait while we load your content...
Please wait while we load your content...