Structure and rheology of dual-associative protein hydrogels under nonlinear shear flow†
Abstract
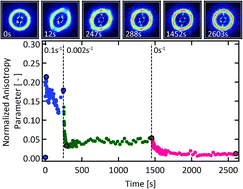
Dual-associative protein di- and triblock copolymers composed of sticker-decorated midblocks and micelle-forming elastin-like polypeptide (ELP) endblocks form shear-thinning, thermoresponsively reinforceable hydrogels that are potentially useful as injectable materials for a variety of applications. Here, the combination of rheological and in situ scattering measurements under shear on these dual-associative gels is employed in order to better understand how block architecture plays a role in controlling microscopic structural rearrangement and the resulting macroscopic mechanical responses. These gels, which form a disordered sphere phase due to endblock aggregation under quiescent conditions with the midblock domains physically crosslinked by protein associations, exhibit both viscoelastic and thixotropic signatures with relative magnitudes dependent upon gel concentration and block architecture. In situ SAXS measurements during flow indicate that these thixotropic responses correspond to the development of ordered domains following start-up of shear. For both architectures, the rate of alignment increases with increasing concentration. However, the rate of domain formation when increasing the temperature from 35 to 50 °C depends on the interplay between thermoresponsive toughening of the endblocks and softening of the coiled-coil domains such that rate of rearrangement decreases in the triblock while it increases in the diblock. Following a step-down in shear flow, structural rearrangement within the samples results in a thixotropic stress response. Upon cessation of flow, gel recovery is characterized by a concentration-dependent restoration of the micellar network over time, with two timescales observed that correspond to two different length scales of network relaxation.


 Please wait while we load your content...
Please wait while we load your content...