The effect of KOH concentration on chemical activation of porous carbon sorbents for carbon dioxide uptake and carbon dioxide–methane selectivity: the relative formation of micro- (<2 nm) versus meso- (>2 nm) porosity†
Abstract
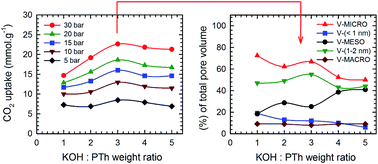
Porous carbon (PC) sorbents are synthesized from polymer precursors mixed with a chemical activation reagent and pyrolised (>500 °C). KOH is known to be the best activator for a wide range of precursors, as it creates PCs with a large surface area (1200–4000 m2 g−1). In order to determine the optimum KOH : polymer ratio for both CO2 adsorption and CO2/CH4 selectivity, we prepared a set of five S-containing porous carbon (SPC) samples from polythiophene (PTh) with increasing KOH : PTh ratio (1 to 5), and investigated CO2 and CH4 uptake measurements on carbonaceous SPC samples up to a pressure limit of 30 bar. The SPCs have been characterised by XPS, SEM, TEM and BET surface area analysis. Although the apparent surface area and total pore volume increased with increasing KOH concentration, the maximum CO2 uptake (5–30 bar) was demonstrated for samples with KOH : PTh = 3. This equates to SPC samples with a surface area and total pore volume of ∼2700 m2 g−1 and 1.5 cm3 g−1, respectively. Greater values for either parameter do not enhance the CO2 uptake, showing that it is not total porosity that is important. SPC samples formed with KOH : PTh = 3 show both a maximum C composition (85%), and a maximum fraction of micro-pores (<2 nm) with a concomitant decrease in meso-pores (>2 nm). KOH : PTh = 3 is also the synthetic condition to maximise CH4 uptake (5–30 bar); however, the optimum CO2/CH4 selectivity occurred with KOH : PTh = 2. This correlates with a different surface area (2200 m2 g−1) and total pore volume (1.2 cm3 g−1) that are required for optimum CO2 uptake. These results suggest that process conditions that lead to high relative microporosity need to be considered rather than the total surface area or pore volume.


 Please wait while we load your content...
Please wait while we load your content...