Reversible mechanical protection: building a 3D “suit” around a T-shaped benzimidazole axle†
Abstract
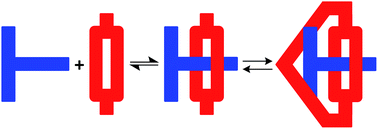
The T-shaped benzimidazolium/crown ether recognition motif was used to prepare suit[1]anes. These novel mechanically interlocked molecules (MIMs) were fully characterized by 1H and 13C NMR spectroscopy, single-crystal X-ray diffraction, UV-vis absorption and fluorescence spectroscopy. By conversion to a suit[1]ane, a simple benzimidazole was shown to be protected from deprotonation by strong base. Moreover, it was demonstrated that this unique three-dimensional encapsulation can be made reversible, thus introducing the concept of “reversible mechanical protection”; a protecting methodology that may have potential applications in synthetic organic chemistry and the design of molecular machinery.
- This article is part of the themed collections: CSC100: Celebrating Canadian Chemistry and International Symposium on Macrocyclic & Supramolecular Chemistry (ISMSC) in conjunction with ISACS


 Please wait while we load your content...
Please wait while we load your content...