Enrichment and fluorogenic labelling of 5-formyluracil in DNA†
Abstract
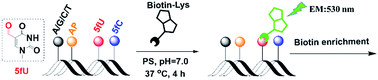
Recently, the detection of natural thymine modified 5-formyluracil has attracted widespread attention. Herein, we introduce a new insight into designing reagents for both the selective biotin enrichment and fluorogenic labelling of 5-formyluracil in DNA. Biotinylated o-phenylenediamine directly tethered to naphthalimide can switch on 5-formyluracil, under physiological conditions, which can then be used in cell imaging after exposure to γ-irradiation. In addition, its labelling property caused the polymerase extension to stop in the 5-formyluracil site, which gave us more information than the fluorescence did itself. The idea of detecting 5-formyluracil might be used in the synthesis of other modified diaminofluoresceins.

 Please wait while we load your content...
Please wait while we load your content...