Continuous microfluidic synthesis of colloidal ultrasmall gold nanoparticles: in situ study of the early reaction stages and application for catalysis†
Abstract
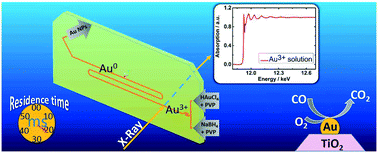
A continuous microfluidic setup was developed to study colloidal synthesis of gold nanoparticles using tetrachloroauric acid as precursor, sodium borohydride as reducing agent and PVP as stabilizer. The setup consists of pressurized vessels that allow pulsation-free flow of reactants and a microfluidic chip with integrated micromixers essential for efficient mixing with small mixing time (2 ms) followed by a meandering microchannel. The microfluidic chip enables recording X-ray absorption spectra (XAS) in situ at different positions along the microchannel at high flow rates approaching turbulent mixing conditions and thus to correlate reaction time with changes in the nanoparticle structure. Significant contributions of oxidized gold could be observed after the first 6 ms of the reaction, whereas after 10 ms principally all gold appeared to be in a metallic state. The nanoparticles obtained were characterized ex situ by various complementary techniques. The resulting nanoparticles had average diameter of 1.0 nm and narrow size distributions compared with those produced in a batch reactor. Depositing the nanoparticles on TiO2 resulted in catalysts with two different Au loadings (0.7 and 1.7 wt% Au/TiO2) which exhibited good CO oxidation activity.


 Please wait while we load your content...
Please wait while we load your content...