Nitrogen doped nanoporous graphene: an efficient metal-free electrocatalyst for the oxygen reduction reaction†
Abstract
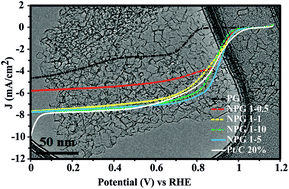
The oxygen reduction reaction (ORR) is an important half reaction, which occurs at the cathode within a fuel cell and limits their range of applications due to slow electrochemical kinetics. To overcome this issue, electrocatalysts are sought, which need to be an alternative to expensive and unsustainable metallic catalysts. Herein we report the synthesis of nitrogen doped nanoporous graphene (NPG), which is a competitive alternative to currently employed metallic catalysts. The NPG is synthesised through a chemical vapour deposition methodology followed by a chemical functionalization step introducing oxygen functional groups (carbonyl and hydroxyl groups), which is then doped with nitrogen via ortho-phenylenediamine (OPDA). The NPG is physiochemically and electrochemically characterised. The NPG demonstrates outstanding electrocatalytic activity towards the ORR in alkaline media proceeding via a favourable 4-electron pathway and is comparable to commercially available platinum–carbon (20%). We demonstrate that the electrochemical activity of the NPG is tailorable such that through increased nitrogen doping the ORR transforms from a 2-electron process to that of the favourable 4-electron process via increasing the proportion of pyridinic nitrogen while the content of graphitic nitrogen remains almost constant. The NPG exhibits excellent electrochemical performance towards the ORR in alkaline media, long-term stability and appropriate methanol crossover as benchmarked to commercialised Pt/C electrodes; this outstanding electrocatalytic activity is related to the high proportion of defects, high porosity and (pyridinic) doping.


 Please wait while we load your content...
Please wait while we load your content...