Binding behaviors and structural characteristics of ternary complexes of β-lactoglobulin, curcumin, and fatty acids†
Abstract
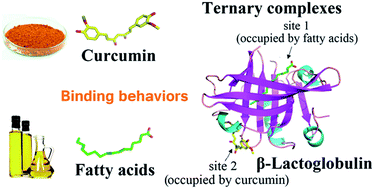
In this study, we evaluated the binding behaviors and structural characteristics of multi-ligand complex of β-lactoglobulin (β-lg) with curcumin and fatty acids. Methods of fluorescence spectroscopy, circular dichroism spectroscopy, dynamic light scattering, Turbiscan stability analysis, HPLC quantification, molecular docking studies, and molecular dynamics simulation were used. The results of fluorescence measurements indicated that β-lg was able to interact with curcumin and fatty acids at the same time, and the presence of fatty acids enhanced the binding constant of curcumin. The formation of ternary complexes with curcumin and fatty acids did not change the secondary structure of β-lg, while this process resulted in a considerable increase in particle size and negative charges compared to the native β-lg (ending up at around 145 nm and −30 mV in the ternary complexes). The loading efficiencies of the fatty acids were 32 ± 2% in β-lg-myristic acid (C14:0)-curcumin and 34 ± 3% in β-lg-palmitic acid (C16:0)-curcumin, and those of curcumin were 58 ± 2% in β-lg-C14:0-curcumin and 57 ± 2% in β-lg-C16:0-curcumin. These findings provide basic information to clarify the binding mechanism for the interactions of β-lg with ligands at multiple sites on the molecular level.


 Please wait while we load your content...
Please wait while we load your content...