Sumalactones A–D, four new curvularin-type macrolides from a marine deep sea fungus Penicillium Sumatrense†
Abstract
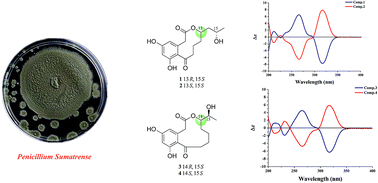
Sumalactones A–D (1–4), four new curvularin-type macrolides, together with two known analogues, curvularin (5) and dehydrocurvularin (6), were isolated from Penicillium Sumatrense, a marine fungus isolated from deep-sea sediments. Sumalactones C (3) and D (4) are unprecedented curvularin-type macrolides bearing a rare 11-membered macrolide skeleton. Their structures were elucidated on the basis of intensive spectroscopic analysis. The absolute configurations of compounds 1–4 were determined by CD spectra and modified Mosher's method. Compound 6 showed significant inhibition activity towards LPS-induced nitric oxide production in RAW 264.7 macrophages with IC50 value of 0.91 μM.


 Please wait while we load your content...
Please wait while we load your content...