Molecular interactions between DOPA and surfaces with different functional groups: a chemical force microscopy study†
Abstract
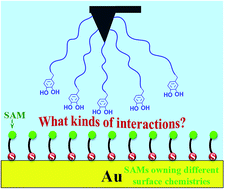
The adhesion of mussel foot proteins (Mfps) to a variety of surfaces has been widely investigated, but the mechanisms behind the mussel adhesion to surfaces with different properties are far from being understood. Most of Mfps contain a significant amount of 3,4-dihydroxyphenylalanine (DOPA) which is considered to be responsible for the strong wet adhesion. In the present work, self-assembled monolayers (SAMs) were prepared as a series of model surfaces with variable functional groups. DOPA-surface interactions were investigated using chemical force microscopy (CFM) for the first time, in which an atomic force microscope (AFM) tip was chemically modified with DOPA terminated groups. The ability of DOPA to adhere to different surfaces with variable wettability was compared, showing that DOPA behaves with the strongest and weakest adhesion to C6H5- and OH-terminated surfaces, respectively. The interaction strength of DOPA at different surfaces does not always increase with the increase of surface wettability, because the hydrophobic interaction does not play a decisive role in DOPA adhering to surfaces. By the use of classical and extended Derjaguin–Landau–Verwey–Overbeek (DLVO) theories, the contribution of non-DLVO forces was isolated. We found out DOPA can adhere to each surface functional group, since DOPA residues containing o-hydroxy or aromatic rings alone can control the adhesion process, and the aromatic ring is oriented perpendicularly or parallel to the surface. This study served as a basis for understanding the relationship between DOPA adhesion mechanisms and different wet surfaces, representing important concepts for the design of bioadhesive materials and anti-adhesion surfaces.


 Please wait while we load your content...
Please wait while we load your content...