Theoretical evaluation of the structure–activity relationship in graphene-based electrocatalysts for hydrogen evolution reactions†
Abstract
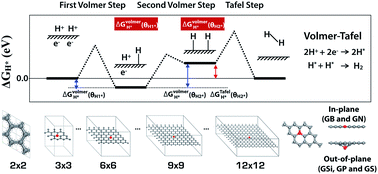
We systematically analyzed the relationship between structure and electrocatalytic activity of heteroatom-doped graphenes (GXs, where G and X represent graphene and the heteroatom dopant) for the hydrogen evolution reaction (HER). We compared the doping effects on the electronic structure and HER activity with the second row elements (B and N) and third row elements (Si, P and S) in the periodic table. In this work, we present evidence that structural deformation and periodic lattice defects play a fundamental role in the HER activity of GXs by adjusting the electronic properties of graphene. We found that graphene doped with third row elements has higher HER activity with out-of-plane structural deformation compared to graphene doped with second row elements, in which graphene tends to maintain its planar structure. In addition, the third row element-doped graphenes (GSi, GP and GS) show an interesting physical regularity described by a simple 3N rule: GXs provide outstanding HER activity with a sustained metallic property when its primitive cell size is a 3N × 3N (N is integral) multiple of that of pure graphene. Therefore, we discuss how a comprehensive understanding of the structure–activity relationship can explain the behavior of new electrocatalytic materials.


 Please wait while we load your content...
Please wait while we load your content...