Electrostatic polypyridine–ruthenium(ii)⋯decatungstate dyads: structures, characterizations and photodegradation of dye†
Abstract
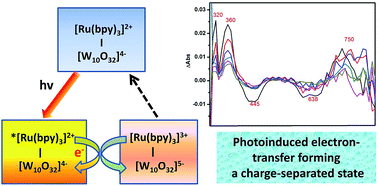
Two hybrids, [Ru(bpy)2(CH3CN)2]2[W10O32]·2CH3CN (RuW-1) and [Ru(bpy)3]2[W10O32] (RuW-2) (bpy = 2,2′-bipyridine), were synthesized by the reaction of (2,2′-dipyridyl)ruthenium complexes and decatungstate, under hydrothermal conditions, and characterized by IR, UV, fluorescence and transient absorption spectroscopy. The influences of the coordination environment of the centre ion, Ru(II), are compared and discussed in detail on the basis of analyzing their photophysical and photochemical properties. Kinetics experiments for the photodegradation of Rhodamine B (RhB) dye were followed with spectrophotometric analysis showing an absorbance decrease at 544 nm resulting from the cleavage of the aromatic ring of RhB. UV spectroscopy indicated that the degradation proceeds with a pseudo-first-order rate constant in the range of 10−3 to 10−2 s−1. These results demonstrate that the hybrids have effective activity and reusability for the photodegradation of RhB.


 Please wait while we load your content...
Please wait while we load your content...