Response surface methodology as a powerful tool to optimize the synthesis of polymer-based graphene oxide nanocomposites for simultaneous removal of cationic and anionic heavy metal contaminants†
Abstract
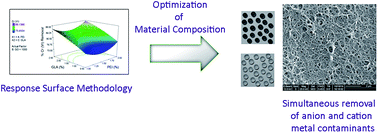
Nanocomposites containing graphene oxide (GO), polyethyleneimine (PEI), and chitosan (CS) were synthesized for chromium(VI) and copper(II) removal from water. Response surface methodology (RSM) was used for the optimization of the synthesis of the CS–PEI–GO beads to achieve simultaneous maximum Cr(VI) and Cu(II) removals. The RSM experimental design involved investigating different concentrations of PEI (1.0–2.0%), GO (500–1500 ppm), and glutaraldehyde (GLA) (0.5–2.5%), simultaneously. Batch adsorption experiments were performed to obtain responses in terms of percent removal for both Cr(VI) and Cu(II) ions. A second-order polynomial equation was used to model the relationship between the synthesis conditions and the adsorption responses. High R2 values of 0.9848 and 0.8327 for Cr(VI) and Cu(II) removal, respectively, were obtained from the regression analyses, suggesting good correlation between observed experimental values and predicted values by the model. The optimum bead composition contained 2.0% PEI, 1500 ppm GO, and 2.08% GLA, and allowed Cr(VI) and Cu(II) removals of up to 91.10% and 78.18%, respectively. Finally, characterization of the structure and surface properties of the optimized CS–PEI–GO beads was carried out using X-ray diffraction (XRD), porosity and BET surface area analysis, scanning electron microscopy (SEM), Fourier transform infrared spectroscopy (FTIR), and X-ray photoelectron spectroscopy (XPS), which showed favorable adsorbent characteristics as given by a mesoporous structure with high surface area (358 m2 g−1) and plenty of surface functional groups. Overall, the synthesized CS–PEI–GO beads were proven to be effective in removing both cationic and anionic heavy metal pollutants.


 Please wait while we load your content...
Please wait while we load your content...