Oxidative bicyclization of N-tethered 1,7-enynes toward polycyclic 3,4-dihydroquinolin-2(1H)-ones via site-selective decarboxylative C(sp3)–H functionalization†
Abstract
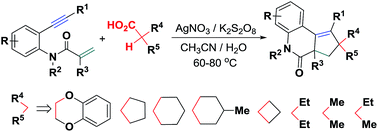
A new Ag-catalyzed oxidative bicyclization of N-tethered 1,7-enynes with alkylcarboxylic acids for forming 41 examples of polycyclic 3,4-dihydroquinolin-2(1H)-ones has been established using readily accessible K2S2O8 as an oxidant. The reaction pathway involves a silver-catalyzed decarboxylation/in situ-generated C-center radical-triggered α,β-conjugated addition/6-exo-dig cyclization/H-abstraction/5-endo-trig cyclization/SET sequence, allowing direct site-selective decarboxylative C(sp3)–H functionalization toward the formation of multiple C–C bonds and rapid construction of complex spiroheterocycles.
- This article is part of the themed collection: Editors’ collection: Catalytic Organic Transformations


 Please wait while we load your content...
Please wait while we load your content...