Identification of DNA single-base mismatches by resistivity of poly(N-isopropylacrylamide)-block-ssDNA copolymer brush films at dual temperatures†
Abstract
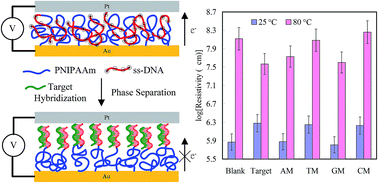
We grafted azido-terminated poly(N-isopropylacrylamide) (PNIPAAm) brushes onto thin gold films on silicon as a bottom electrode. A probe of single-stranded DNA (ssDNA) presenting a 4-pentynoic acid succinimidyl ester unit was grafted onto the azido-terminated PNIPAAm brushes through a click reaction, resulting in block copolymer brushes. The PNIPAAm-b-ssDNA copolymer brushes formed homogeneous complexes stabilized via bio-multiple hydrogen bonds (BMHBs), which enhanced proton transfer and thereby decreased the resistivity of the structures. The homogeneous complex state of the PNIPAAm-b-ssDNA copolymer brushes transformed into a phase-separated state after hybridization with 0.5 pg nL−1 of its target DNA, which resulted from competition between the BMHBs and complementary HBs; this phase transformation of the PNIPAAm and probe segments inhibited proton transfer and significantly increased the resistivity. Furthermore, hybridization with mismatched DNA sequences generated sufficient “proton leakage” to decrease the resistivity at temperatures below the lower critical solution temperature (LCST), especially for adenine and guanine units. Sequences with thymine and cytosine mismatches could be distinguished from the target at temperatures above the LCST due to disruption of the BMHBs at increased temperatures (80 °C). The ability to detect label-free DNA and recognize sequence mismatches suggests the potential use of these novel materials in bioelectronics.


 Please wait while we load your content...
Please wait while we load your content...