REBaCo2O5+δ (RE = Pr, Nd, and Gd) as promising oxygen electrodes for intermediate-temperature solid oxide electrolysis cells
Abstract
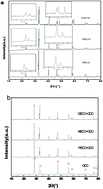
Double-perovskite REBaCo2O5+δ (RE = Pr, Nd, and Gd) oxides were synthesized and evaluated as oxygen electrodes for intermediate-temperature solid oxide electrolysis cells (IT-SOECs). The crystal structures, thermal stability properties, electrical conductivities, and electrochemical performances of the REBaCo2O5+δ materials were investigated systematically. Among the three oxides, PrBaCo2O5+δ (PBCO) has the highest electrical conductivity of 645 S cm−1 at 600 °C. The electrical conductivity decreases as the ionic radius of the RE decreases, which is related to the increase in oxygen vacancy concentration. NdBaCo2O5+δ (NBCO) has the lowest polarization resistance of 0.95 Ω cm2 among the three oxides at 650 °C, which is mainly because NBCO has a better balance between thermal expansion compatibility and conductivity than PBCO and GdBaCo2O5+δ (GBCO). For electrochemical performance in SOECs, NBCO has the smallest current/voltage loss; at a voltage of 1.2 V, the power density of NBCO is 1.58 W cm−2 at 800 °C. Even at 700 °C, a maximum power density of 0.857 W cm−2 can still be achieved by NBCO. These results suggest that NBCO is a novel and promising candidate oxygen electrode material for IT-SOECs.


 Please wait while we load your content...
Please wait while we load your content...