Low-temperature solution synthesis of CuO/Cu2O nanostructures for enhanced photocatalytic activity with added H2O2: synergistic effect and mechanism insight†
Abstract
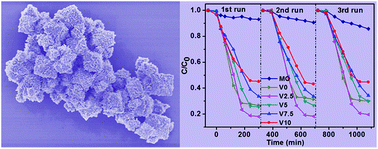
CuxO (CuO, CuO/Cu2O, and Cu2O) nanostructures have been controllably synthesized through a facile low-temperature solution method. The morphologies and compositions of CuxO nanostructures were well controlled by tuning the reductant amount of hydroxylamine hydrochloride, which experienced the transformation of nanosheets to octahedrons as well as the phase change of CuO to Cu2O. The as-grown samples showed photodegradation selectivity to MO (maximum photocatalytic efficiency of 52% for methyl orange (MO) and 16% for rhodamine B (RhB) after 180 min photodegradation) and different photocatalytic activities in the absence or presence of H2O2 (62% without H2O2 and 82% with H2O2). The morphological transformations of as-grown samples were observed after the photocatalytic measurement in the presence of H2O2. The structural and morphological features after photodegradation were studied by XPS, SEM, and TEM investigations, revealing the possible mechanism of the as-prepared samples whereby the photodegradation of organic dyes occurred on the surface with respect to the adsorption ability, structure, and morphology of CuxO. In addition, H2O2 played an important role in the photodegradation of organic dyes.


 Please wait while we load your content...
Please wait while we load your content...