A highly efficient heterogeneous copper-catalyzed chlorodeboronation of arylboronic acids leading to chlorinated arenes
Abstract
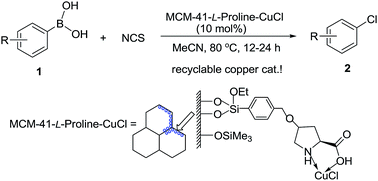
A highly efficient heterogeneous copper-catalyzed chlorodeboronation of arylboronic acids with inexpensive N-chlorosuccinimide (NCS) was achieved in MeCN in the presence of 10 mol% of L-proline-functionalized MCM-41-immobilized copper(I) complex [MCM-41-L-proline-CuCl] under mild conditions, yielding a variety of aryl chlorides in excellent yields. This method proved to be tolerant of a broad range of functional groups and particularly useful for the conversion of electron-deficient arylboronic acids to aryl chlorides, a transformation that is inefficient without copper catalysis. This heterogeneous copper catalyst can be recovered by a simple filtration of the reaction solution and recycled for at least 10 times without any decreases in activity.

 Please wait while we load your content...
Please wait while we load your content...