Diverse exploitation of Brønsted acid catalysts – paving the way for simple access to enantioenriched amines
Abstract
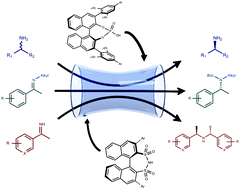
Recent advances in the catalytic enantioselective synthesis of primary and secondary amines are highlighted. These reactions are promoted by chiral Brønsted acid catalysts, which maintain their activity even in the presence of basic amine products. More in detail, a conceptually new approach to the kinetic resolution of racemic amines, based on a condensation process, and two transfer hydrogenation reactions of rarely employed N-alkyl and N–H ketimines with Hantzsch esters, are summarised in this article.


 Please wait while we load your content...
Please wait while we load your content...