A facile approach to ketones via Pd-catalyzed sequential carbonylation of olefins with formic acid†
Abstract
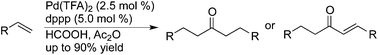
An effective palladium-catalyzed sequential carbonylation of olefins with formic acid is described. Ketones and the corresponding α,β-enones can be obtained with up to 90% yield. The reaction process is operationally simple and requires no toxic CO.
- This article is part of the themed collection: Organic Chemistry Frontiers HOT articles for 2017

 Please wait while we load your content...
Please wait while we load your content...