Charge mobility enhancement for diketopyrrolopyrrole-based conjugated polymers by partial replacement of branching alkyl chains with linear ones†
Abstract
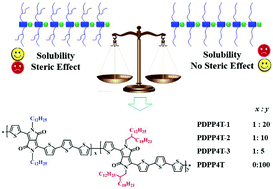
Conjugated D–A polymers with high charge mobilities have received increasing attention in recent years. In this paper, we report a simple, but efficient approach to improve the charge mobilities of conjugated polymers by partial replacement of branching alkyl chains with linear ones. With this strategy in mind, we designed and investigated three new DPP-quaterthiophene terpolymers PDPP4T-1, PDPP4T-2 and PDPP4T-3 containing both linear (n-dodecane) and branching (2-decyltetradecyl) alkyl chains with different ratios. On the basis of GIWAXS data, interchain packing order is improved for terpolymers PDPP4T-1, PDPP4T-2 and PDPP4T-3, in comparison with PDPP4T with only branching alkyl chains. The incorporation of linear alkyl chains can affect the interchain packing mode, and PDPP4T-3 with more linear alkyl chains among the three terpolymers adopts the edge-on chain arrangement on the substrate. Field effect transistor measurements indicate that PDPP4T-1, PDPP4T-2 and PDPP4T-3 exhibit higher hole mobilities than PDPP4T. Moreover, the hole mobilities increase in the following order: PDPP4T-1 < PDPP4T-2 < PDPP4T-3 by increasing the content of linear alkyl chains in these terpolymers. The thin film mobility of PDPP4T-3 can reach 6.1 cm2 V−1 s−1, which can be further increased to 9.1 cm2 V−1 s−1 after incorporating NMe4I.
- This article is part of the themed collections: Molecular Materials and Devices and Materials Chemistry Frontiers HOT articles for 2017


 Please wait while we load your content...
Please wait while we load your content...