3D coffee stains†
Abstract
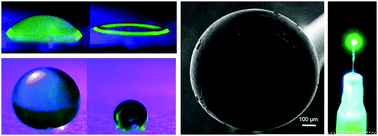
When a liquid droplet (e.g., coffee, wine, etc.) is splattered on a surface, the droplet dries in a ring-shaped stain. This widely observed pattern in everyday life occurs due to the phenomenon known as a coffee stain (or coffee ring) effect. While the droplet dries, the capillary flow moves and deposits the particles toward the pinned edges, which shows a 2D ring-like structure. Here we demonstrate the transition from a 2D to a 3D coffee stain that has a well-defined and hollow sphere-like structure, when the substrate surface is switched from hydrophilic to superhydrophobic. The 3D stain formation starts with the evaporation of the pinned aqueous colloidal droplet placed on a superhydrophobic surface that facilitates the particle flow towards the liquid–air interface. This leads to spherical skin formation and a cavity in the droplet. Afterwards the water loss in the cavity due to pervaporation leads to bubble nucleation and growth, until complete evaporation of the solvent. In addition to the superhydrophobicity of the surface, the concentration of the solution also has a significant effect on 3D coffee stain formation. Advantageously, 3D coffee stain formation in a pendant droplet configuration enables the construction of all-protein lasers by integrating silk fibroin with fluorescent proteins. No tools, components and/or human intervention are needed after the construction process is initiated; therefore, 3D coffee-stains hold promise for building self-assembled and functional 3D constructs and devices from colloidal solutions.
- This article is part of the themed collection: 2017 Emerging Investigators by MCF


 Please wait while we load your content...
Please wait while we load your content...