Highly efficient thermally activated delayed fluorescence materials with reduced efficiency roll-off and low on-set voltages†
Abstract
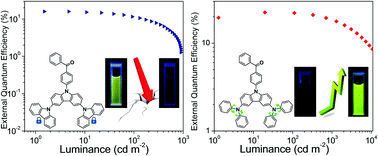
Two carbazoles or diphenylamines were combined with carbazole to obtain two branch-shaped donor units and then connected to a benzophenone unit, acquiring two donor–acceptor (D–A) type thermally activated delayed fluorescence (TADF) materials named CCDC and CCDD. CCDC and CCDD demonstrate opposing phenomena in a THF/H2O mixed solvent despite their structural similarity. The photoluminescence (PL) of CCDC drops drastically as the water proportion goes up, while the PL of CCDD increases with an increasing water ratio. The lower PL emission of CCDD in good solvents may be attributed to the vibration and rotation of the phenyl rings in the diphenylamine donors, and the PL emission increases when the intramolecular movement is restricted. However, PL quantum yield (PLQY) tests and device performances indicate that both materials experience aggregation-caused quenching (ACQ) to some extent in a solid state film. This is because when intramolecular vibration and rotation is restrained in the solid state, intermolecular stacking still hampers the fluorescence emission of CCDD. Also, the materials perform well in doped devices. A maximal external quantum efficiency (EQE) of 15.9% is achieved in blue-emitting devices with CCDC as the emitter. Green-emitting devices where CCDD is utilized as the emitter perform well regarding their overall properties. Not only do their maximal EQEs exceed 22%, but they also achieve on-set voltages as low as 2.6 V with a 30% doping concentration. Moreover, the efficiency roll-off of the CCDD-based devices is well controlled. An EQE of nearly 20% is maintained at a luminance of 1000 cd m−2.
- This article is part of the themed collections: Molecular Materials and Devices and MCF Editors’ Recommendation


 Please wait while we load your content...
Please wait while we load your content...