Catalytic aerial oxidation of 5-hydroxymethyl-2-furfural to furan-2,5-dicarboxylic acid over Ni–Pd nanoparticles supported on Mg(OH)2 nanoflakes for the synthesis of furan diesters†
Abstract
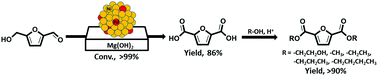
Efficient catalytic aerial oxidation of biomass-derived 5-hydroxymethyl-2-furfural (5-HMF) to the industrially important furan-2,5-dicarboxylic acid (FDCA) was achieved over bimetallic M0.90–Pd0.10 (M = Ni, Co or Cu) alloy nanoparticles supported on in situ prepared Mg(OH)2 nanoflakes. In comparison to Co- and Cu-based bimetallic nanoparticles, Ni0.90Pd0.10/Mg(OH)2 exhibited superior catalytic performance. Catalytic reactions performed using Ni1−xPdx/Mg(OH)2 (x = 0.10 to 1) and a physical mixture of Ni/Mg(OH)2 and Pd/Mg(OH)2 revealed that the significant synergistic cooperation between Ni and Pd plays a crucial role in the observed high catalytic activity of Ni0.90Pd0.10/Mg(OH)2 for the oxidation of 5-HMF to FDCA. Experiments performed with Ni0.90Pd0.10 on different supports (SiO2, Al2O3, ZnO and Mg(OH)2) revealed that the basicity of the support is advantageous for efficient oxidation of 5-HMF, and it avoids the use of an external base additive. Moreover, the Ni0.90Pd0.10/Mg(OH)2 catalytic system can be scaled to gram-level oxidation of 5-HMF to FDCA. Subsequently, the synthesized FDCA was employed for the synthesis of various furan diesters (bis(2-hydroxyethyl), dimethyl, diethyl, dipropyl and dibutyl furan-2,5-dicarboxylate) as precursors for biomass-derived plastics. Moreover, structural and chemical identification of the highly active Ni0.90Pd0.10/Mg(OH)2 was established by P-XRD, SEM, TEM, EDS, elemental mapping and ICP-AES analysis.


 Please wait while we load your content...
Please wait while we load your content...