Metallopolymers from direct polymerization of functionalized cobalt chalcogenide clusters and thiophene comonomers†
Abstract
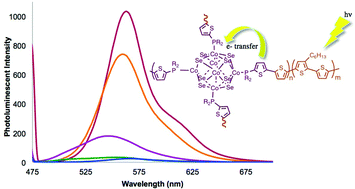
Semiconducting quantum dots and functionalized nanoparticles have been limited in their use within hybrid photovoltaic devices due to their poor percolation into the organic polymer matrix. The copolymerization of a functionalized cobalt chalcogenide cluster (Co6Se8(P(Ph)2(C4H2SBr))6) (1) with 2,5-bistrimethylstannylthiophene and 2,5-dibromo-3-hexylthiophene results in a type III metallopolymer. The ratio of cluster to the other comonomers was varied to yield three new copolymers, poly-cluster-co-thiophene-co-3-hexylthiopehene, PCLTHTa–c. Structural characterization was performed with NMR, GPC and UV-visible spectroscopy. DFT calcuations and experimental results were used to determine the impact of attachment on the polymer chain conjugation. Charge transfer from the polymer to the cluster moieties is observed in all of the cluster–thiophene copolymers. Increased charge transfer is observed with increased cluster incorporation. Fluorescence quenching experiments comparing simple cluster–polymer mixtures to the polymerized clusters indicate that covalent attachment of the cluster into the polymer chain can result in improved charge transfer. Optical and electronic characterization of thin films of PCLTHTa–c supports formation of uniform films and suggests that increasing polymer chain lengths lead to increased order in the solid-state.

 Please wait while we load your content...
Please wait while we load your content...