Structure–cytotoxicity relationship of drug-initiated polymer prodrug nanoparticles†
Abstract
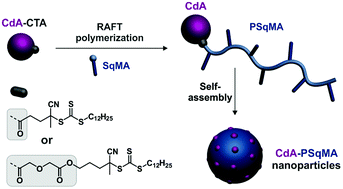
Cladribine (CdA)-based polymer prodrug nanoparticles were obtained by “drug-initiated” RAFT polymerization of squalenyl methacrylate (SqMA) from two different CdA-bearing chain transfer agents (CTAs) to evaluate the influence of the nature of the linkage (a single linear ester or a linear ester linked to a diglycolate linker) between CdA and the CTA, and the bulkiness of the polymer, on the drug release and the biological performance. The different conjugates exhibited the expected structures and were well-controlled as shown by 1H NMR and SEC (Đ = 1.12–1.27). The resulting nanoparticles obtained after nanoprecipitation in aqueous solution were narrowly dispersed with average diameters ranging from 69 to 112 nm and strongly negative surface charges (ζ = −51 to −61 mV). By varying the Mn, drug loadings ranged from 6.4 to 10.5 wt%, leading to significant cytotoxicity in vitro on mouse lymphocytic leukemia (L1210) cells. These results were combined with the already published literature to extract a preliminary structure–cytotoxicity relationship for CdA-based polymer prodrug nanoparticles, involving the nature of the linkage between CdA and the polymer, the nature of the polymer and the Mn.
- This article is part of the themed collection: Pioneering Investigators


 Please wait while we load your content...
Please wait while we load your content...