Triple-stimuli responsive polymers with fine tuneable magnetic responses†
Abstract
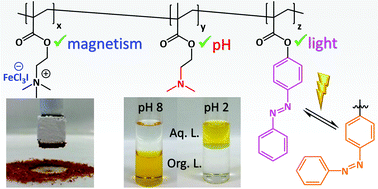
The formation of multi-stimuli responsive polymers exhibiting magnetic, pH and light sensitivity is reported. After quaternization of tertiary amines along poly(2-(N,N-dimethylamino) ethyl methacrylate) (PDMAEMA) with varied alkyl and benzyl halides, a series of tetrahalogenoferrate(III) salts were obtained by anion metathesis, conferring tailored and high magnetic responses. Experimental and theoretical Raman analyses allowed one to distinguish between the different magnetic anions present in the materials. This strategy was then implemented to the formation of multi-responsive compounds, by using random copolymers that incorporate various amounts of DMAEMA (∼40, 65 and 80%) and diazobenzene units. Partial or total quaternization of the dimethylamino end groups (10, 50 and 100%) followed by magnetic anion exchange afforded the magnetic-responsive copolymers, as evidenced by their magnetic susceptibility. Free ternary amino groups and diazobenzene moieties further brought pH and light sensitivity, giving rise to triple-stimuli responsive materials. This simple and rapid procedure offers the opportunity to fine tune the magnetic properties of remotely controlled smart devices.


 Please wait while we load your content...
Please wait while we load your content...