Rational design of soluble and clickable polymers prepared by conventional free radical polymerization of acetylene-functionalized acrylate†
Abstract
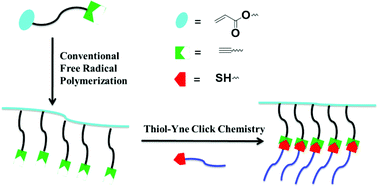
The radical polymerization of acetylene-bearing acrylates to linear clickable polymers represents a synthetic challenge because acetylenes are too reactive to avoid chain branching or cross-linking. Herein, we report a methodology for the synthesis of acetylene-functionalized clickable polymers via conventional free radical polymerization of a novel acrylate bearing tertiary acetylene, 1-ethynylcyclohexyl acrylate (ECA). This methodology allows the acetylene-functionalized polymers to be prepared by conventional free radical polymerizations without the need to protect the acetylene group before polymerization and without the help of an ATRP/RAFT agent. The characteristics of ECA in free radical polymerization and copolymerization including bulk polymerization and conventional solution polymerization at varied concentrations and reaction times were studied. High molecular weight acetylene-functionalized polymers were prepared and were characterized by SEC, 1H-NMR, FTIR and DSC. When ECA was coploymerized with methyl acrylate, as the feed ratio of ECA increased, the rate of the polymerization reaction and the molecular weight of the copolymer decreased while the molecular weight distribution (PDI) slightly increased. The glass transition temperature of PECA was found to be 110 °C by DSC. The acetylene-decorated polymer chains are thermally cross-linkable and photochemically thiol–yne clickable.


 Please wait while we load your content...
Please wait while we load your content...