Synthetic modulation of micro- and mesopores in polycyanurate networks for adsorptions of gases and organic hydrocarbons†
Abstract
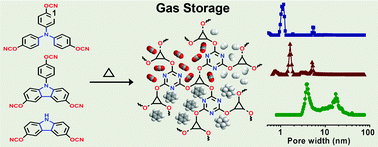
Three new cyanate monomers tris(4-cyanatophenyl)amine, 9-(4′-cyanatophenyl)-3,6-dicyanatocarbazole and 3,6-dicyanatocarbazole with varied geometrical shape, size and number of functional groups were designed and synthesized, which were then thermally polymerized, respectively, to create three micro- and mesoporous polycyanurate networks (PCN-TA, PCN-TC and PCN-DC). The resultant polymers show tunable pore sizes from 1.17 nm to 17.3 nm and BET surface areas from 393 to 721 m2 g−1. Among the three samples, PCN-TA with the smallest pore size possesses the largest uptake for H2 (1.13 wt%, 77 K/1.0 bar) and CO2 (11.0 wt%, 273 K/1.0 bar), and meanwhile displays the highest CO2/CH4 adsorption selectivity up to 24 (IAST method). The gas adsorption/separation properties are studied and explained in terms of the variations of porous and chemical structures of polycyanurate networks as well as the critical temperature, kinetic diameter and polarity of gas molecules. In addition, it is interesting to observe that PCN-TA can uptake 100.9 wt% benzene at 298 K and P/P0 = 0.9. Moreover, its adsorption capacity for benzene is about five times as high as that of cyclohexane, exhibiting promising application in selective adsorption of benzene from the benzene/cyclohexane mixture.


 Please wait while we load your content...
Please wait while we load your content...