Controlling the folding of conjugated polymers at the single molecule level via hydrogen bonding†
Abstract
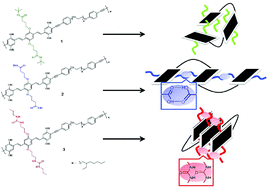
In this manuscript, we report a design strategy to control polychromophore polymer folding at the single molecule level through hydrogen-bonding (H-bonding) interactions. Polymers composed of bis(2-ethylhexyl)-p-phenylene vinylene (BEH-PPV) oligomers (trimers) containing H-bonding capable side-chains (e.g., carboxylic acid or urea units) were prepared by copolymerization with flexible linkers. Control polymers with masked H-bonding side-chains (e.g., t-butyl ester) were also prepared. Different polymer folding schemes are proposed including random or non-folding for the control polymers, and side-on and face-on geometries for the carboxylic acid and urea motifs, respectively. Single molecule excitation polarization spectroscopy demonstrates that the highest folding order is achieved with the urea containing side-chains and isotropic folding is found in polymers that do not contain H-bonding units. We propose that the effects of the chromophore conformational geometries and the type of H-bonding are additive and lead to highly aligned urea-containing polymers. In addition, an examination of the individual polymers’ spectral signatures via single molecule spectroscopy showed an overall red-shift of ∼0.06 eV in the 0–0 origin peak for the urea-containing polymer compared to the other two polymer systems. This spectral shift is attributed to planarization of the backbone and further supports the presence of a highly ordered urea-containing polymer structure. Utilization of this H-bonding inclusion synthetic strategy to control polymer secondary structure could provide important design elements for future functional material design.


 Please wait while we load your content...
Please wait while we load your content...