Optimization of a synthetic receptor for dimethyllysine using a biphenyl-2,6-dicarboxylic acid scaffold: insights into selective recognition of hydrophilic guests in water†
Abstract
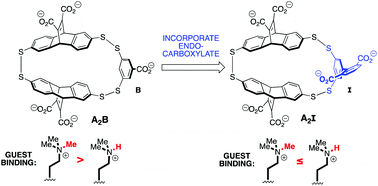
In the design of small molecule receptors for polar guests, much inspiration has been taken from proteins that have adapted effective ways to selectively bind polar molecules in aqueous environments. Nonetheless, molecular recognition of hydrophilic guests in water by synthetic receptors remains a challenging task. Here we report a new synthetic receptor, A2I, with improved affinity and selectivity for a biologically important polar guest, dimethyllysine (Kme2). A2I was prepared via redesign of a small molecule receptor (A2B) that preferentially binds trimethyllysine (Kme3) using dynamic combinatorial chemistry (DCC). We designed a new biphenyl-2,6-dicarboxylate monomer, I, with the goal of creating a buried salt bridge with Kme2 inside a synthetic receptor. Indeed, incorporation of I into the receptor A2I resulted in a receptor with 32-fold enhancement in binding affinity, which represents the highest affinity receptor for Kme2 in the context of a peptide to date and is tighter than most Kme2 reader proteins. It also exhibits a ∼2.5-fold increase in preference for Kme2 vs. Kme3 relative to the parent receptor, A2B. This work provides insight into effective strategies for binding hydrophilic, cationic guests in water and is an encouraging result toward a synthetic receptor that selectively binds Kme2 over other methylation states of lysine.
- This article is part of the themed collection: Celebrating excellence in research: women of organic chemistry


 Please wait while we load your content...
Please wait while we load your content...