Recognition of mixed-sequence DNA using double-stranded probes with interstrand zipper arrangements of O2′-triphenylene- and coronene-functionalized RNA monomers†
Abstract
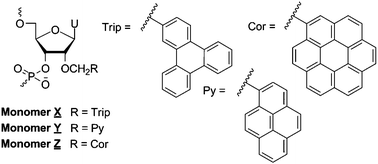
Development of hybridization-based probes that enable recognition of specific mixed-sequence double-stranded DNA (dsDNA) regions is of considerable interest due to their potential applications in molecular biology, biotechnology, and medicine. We have recently demonstrated that nucleic acid duplexes with +1 interstrand zipper arrangements of intercalator-functionalized nucleotides such as 2′-O-(pyren-1-yl)methyl RNA monomers are inherently activated for recognition of mixed-sequence dsDNA targets, including chromosomal DNA. In the present work, we follow up on our previous structure–activity relationship studies and explore if the dsDNA-recognition efficiency of these so-called Invader probes can be improved by using larger intercalators than pyrene. Oligodeoxyribonucleotides modified with 2′-O-(triphenylen-2-yl)methyl-uridine monomer X and 2′-O-(coronen-1-yl)methyl-uridine monomer Z form extraordinarily stabilized duplexes with complementary DNA (ΔTm's per modification of up to 13 °C and 20 °C, respectively). Invader probes based on X- and Z-monomers are shown to recognize model dsDNA targets with exceptional binding specificity, but are less efficient than reference probes modified with 2′-O-(pyren-1-yl)methyl-uridine monomer Y. The insight from this study will inform further optimization of Invader probes.
- This article is part of the themed collection: Nucleic Acid Modifications


 Please wait while we load your content...
Please wait while we load your content...