Positive zeta potential of nanodiamonds†
Abstract
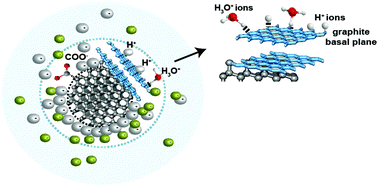
In this paper, the origin of positive zeta potential exhibited by nanodiamond particles is explained. Positive zeta potentials in nano-structured carbons can be explained by the presence of graphitic planes at the surface, which leave oxygen-free Lewis sites and so promotes the suppression of acidic functional groups. Electron Microscopy and Raman Spectroscopy have been used to show that positive zeta potential of nanodiamond is only exhibited in the presence of sp2 carbon at the surface.


 Please wait while we load your content...
Please wait while we load your content...