Determination of the nano-scaled contact area of staphylococcal cells†
Abstract
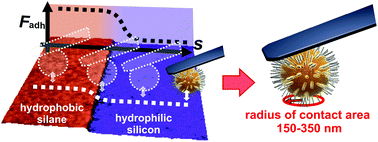
Bacterial adhesion is a crucial step during the development of infections as well as the formation of biofilms. Hence, fundamental research of bacterial adhesion mechanisms is of utmost importance. So far, less is known about the size of the contact area between bacterial cells and a surface. This gap will be filled by this study using a single-cell force spectroscopy-based method to investigate the contact area between a single bacterial cell of Staphylococcus aureus and a solid substrate. The technique relies on the strong influence of the hydrophobic interaction on bacterial adhesion: by incrementally crossing a very sharp hydrophobic/hydrophilic interface while performing force–distance curves with a single bacterial probe, the bacterial contact area can be determined. Assuming circular contact areas, their radii – determined in our experiments – are in the range from tens of nanometers to a few hundred nanometers. The contact area can be slightly enlarged by a larger load force, yet does not resemble a Hertzian contact, rather, the enlargement is a property of the individual bacterial cell. Additionally, Staphylococcus carnosus has been probed, which is less adherent than S. aureus, yet both bacteria exhibit a similar contact area size. This corroborates the notion that the adhesive strength of bacteria is not a matter of contact area, but rather a matter of which and how many molecules of the bacterial species’ cell wall form the contact. Moreover, our method of determining the contact area can be applied to other microorganisms and the results might also be useful for studies using nanoparticles covered with soft, macromolecular coatings.


 Please wait while we load your content...
Please wait while we load your content...