Enhanced hydrogen adsorption on graphene by manganese and manganese vanadium alloy decoration
Abstract
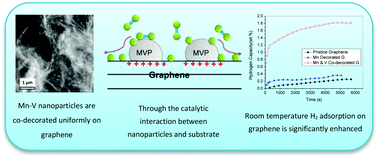
In this work, two kinds of novel manganese decorated (G + Mn) and manganese-vanadium co-decorated (G + MnV) graphene composites are synthesized by in situ wet chemical reduction, and their hydrogen storage properties and microstructures are characterized by Sievert-type adsorption apparatus, BET, SEM, TEM/STEM, EDX and EELS. Compared with pristine graphene, Mn decoration marginally increases the hydrogen adsorption capacity of graphene at room temperature and 4 MPa hydrogen pressure from 0.25 wt% to 0.36 wt%. On the other hand, the co-decoration of Mn and V increases the room temperature hydrogen storage capacity of graphene significantly to 1.81 wt% under 4 MPa hydrogen pressure, which is 1.56 wt% higher than the capacity of pristine graphene. The microstructures and valence states of the decorated Mn and Mn–V nanoparticles are investigated by TEM, EDX and EELS analyses, and strong interactions between the decorated nanoparticles and graphene are observed. Based on the results from structural analyses, potential enhancement mechanisms are suggested in terms of the catalytic effects of nanoparticles on graphene hydrogen adsorption. Given the relatively low cost of Mn and V metals compared to noble metals such as Pd, Pt and Au, these results demonstrate a low cost and effective way to significantly enhance the room temperature hydrogen adsorption properties of graphene for potential hydrogen storage applications.


 Please wait while we load your content...
Please wait while we load your content...