Mechanism of α-synuclein translocation through a VDAC nanopore revealed by energy landscape modeling of escape time distributions
Abstract
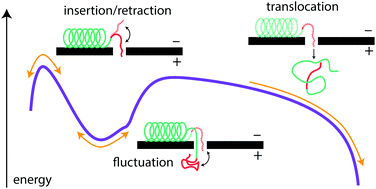
We probe the energy landscape governing the passage of α-synuclein, a natural “diblock copolymer”-like polypeptide, through a nanoscale pore. α-Synuclein is an intrinsically disordered neuronal protein associated with Parkinson's pathology. The motion of this electrically heterogeneous polymer in the β-barrel voltage-dependent anion channel (VDAC) of the mitochondrial outer membrane strongly depends on the properties of both the charged and uncharged regions of the α-synuclein polymer. We model this motion in two ways. First, a simple Markov model accounts for the transitions of the channel between the states of different occupancy by α-synuclein. Second, the detailed energy landscape of this motion can be accounted for using a drift-diffusion framework that incorporates the α-synuclein binding energy and the free energy cost of its confinement in the VDAC pore. The models directly predict the probability of α-synuclein translocation across the mitochondrial outer membrane, with immediate implications for the physiological role of α-synuclein in regulation of mitochondrial bioenergetics. Time-resolved measurements of the electrical properties of VDAC occupied by α-synuclein reveal distinct effects of the motion of the junction separating the differently charged regions of the polymer.

 Please wait while we load your content...
Please wait while we load your content...