Fluorescent carbon quantum dots chemosensor for selective turn-on sensing of Zn2+ and turn-off sensing of Pb2+ in aqueous medium and zebrafish eggs
Abstract
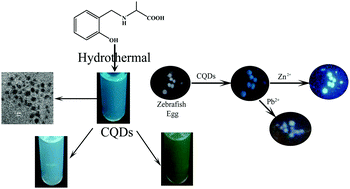
Fluorescent carbon quantum dots (CQDs) synthesized using an amino acid-based phenolic chelating ligand, 2-(2-hydroxybenzylamino)propanoic acid, via a hydrothermal method showed selective turn-on fluorescence for Zn2+ and turn-off for Pb2+ in aqueous medium. High-resolution transmission electron microscopy analysis revealed uniformly dispersed spherical CQDs (3–5 nm). Fourier transform infrared and X-ray photoelectron spectroscopy studies confirmed carboxyl, hydroxy and amine-based surface functional groups. The CQDs exhibited good fluorescence intensity at 488 nm as well as excitation wavelength-dependent fluorescence. Interestingly, Zn2+ addition selectively blue shifted the fluorescence from 488 to 460 nm with a broad fluorescence, which led to white emission in the solution. In contrast, Pb2+ addition substantially quenched the fluorescence with a red shift in λmax. Thus, the CQDs showed selective sensing of Zn2+ and Pb2+ ions in aqueous media with completely distinguishable response. Concentration-dependent studies suggested that the CQDs can detect down to μM concentrations of Zn2+ and Pb2+ ions. Interference studies revealed good selectivity for Zn2+ and Pb2+ ions in the presence of other metal cations. Importantly, the fluorescence sensing of both Zn2+ and Pb2+ ions was performed in zebrafish eggs to demonstrate the biological application of the synthesized CQDs.


 Please wait while we load your content...
Please wait while we load your content...