Synthesis process, size and composition effects of spherical Fe3O4 and FeO@Fe3O4 core/shell nanoparticles
Abstract
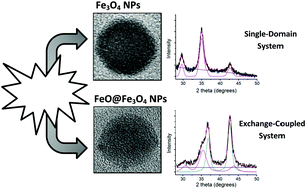
In this work, we investigate the size, composition and magnetic behavior of a series of iron oxide nanoparticles prepared by means of high temperature decomposition of an iron oleate precursor. Different synthesis conditions, such as gas atmosphere, precursor ratio and heating rate were tested to obtain a direct correlation between the final sample structure and the varied parameter. The synthesis products were characterized by X-ray diffraction, transmission electron microscopy and small-angle X-ray scattering, respectively. We studied six samples with rather narrow size distribution and mean diameters from 8 nm to 16 nm. The particles with diameter below 11 nm were found to be spinel-type, monocrystalline, and their magnetic response can be ascribed to a single domain framework. On the other hand, two-phase core–shell FeO@Fe3O4 of mean sizes of 15 nm and 16 nm were obtained by increasing the amount of oleic acid and the heating rate. The magnetic behavior of these samples exhibits interesting interface features, related to the exchange coupling phenomenon between the FeO and Fe3O4. We discuss how the different synthesis conditions may lead to the presence of this FeO phase, and how the core–shell configuration and other structural features affect the macroscopic magnetic behavior.


 Please wait while we load your content...
Please wait while we load your content...