Exonuclease III-aided autonomous cascade signal amplification: a facile and universal DNA biosensing platform for ultrasensitive electrochemical detection of S. typhimurium
Abstract
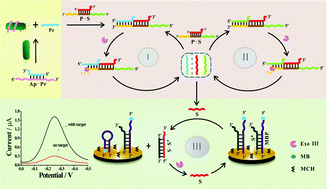
In this study, a novel electrochemical biosensor based on exonuclease III (Exo III)-aided autonomous cascade signal amplification for ultrasensitive and highly specific detection of Salmonella typhimurium (S. typhimurium) has been reported. Herein, two duplex DNA probes were ingeniously designed. An Ap–Pr duplex probe was constructed as a recognition element by hybridizing an S. typhimurium aptamer (Ap) with a primer (Pr). A P–S duplex probe has been designed to contain a protruding DNA fragment at 5′-termini that has an identical sequence with Pr (the secondary Pr), a complementary duplex DNA that is used for blocking a signal-triggered probe (S), and an overhung nucleic acid segment at 3′-end that can anneal with Pr and form double-stranded DNA with a blunt 3′-terminus, thus initiating Exo III-aided amplification reaction. Upon sensing of the target S. typhimurium, the specific recognition of S. typhimurium and aptamer releases Pr, which anneals with the P–S duplex and triggers the Exo III cleavage reaction, accompanied by release of Pr, S, and secondary Pr. After this, the released Pr and secondary Pr anneal with other P–S duplex, which in turn triggers the Exo III-assisted cycling cleavage. Thus, large amounts of S are liberated, which hybrids with S* (DNA probe complementary to S); thereby, 5′-methylene blue (MB)-labelled DNA probe (MBP) folds into a hairpin structure with 5′-terminus close to electrode surface for efficient electronic transfer. Moreover, the S–S* hybrid duplex is specifically hydrolyzed by Exo III to liberate S, which accordingly results in the cycling cleavage of S*. Therefore, numerous MBP transform into a hairpin structure with electroactive MB close to the electrode surface; this generates significantly amplified electrochemical signal towards S. typhimurium detection. The present developed strategy exhibited high sensitivity towards S. typhimurium with a detection limit of 2.8 × 10 cfu mL−1. Furthermore, the proposed autonomous cascade signal amplification strategy can be easily extended for detection towards a wide spectrum of analytes including nucleic acid, proteins, and small molecules possessing their corresponding aptamers. Therefore, the developed Exo III-aided autonomous cascade signal amplification strategy might indeed provide a facile and universal tool for bioanalysis and clinical diagnosis.


 Please wait while we load your content...
Please wait while we load your content...