Rotaxanes composed of dibenzo-24-crown-8 and macrocyclic transition metal complexing tetraimine units†
Abstract
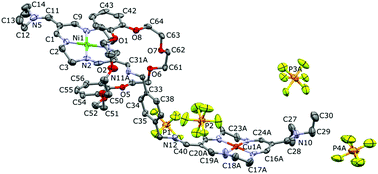
We present the structures of new rotaxanes. They are composed of dibenzo-24-crown-8 and an axle containing two tetraazamacrocyclic units coordinating the same, or different, metal ions (Ni or/and Cu). These units are linked with the m- and p-xylylene bridges. The structures of these compounds were confirmed by single crystal X-ray diffraction, NMR, and other spectroscopic methods. Rigid linkers between the macrocyclic units allow control of the axle length and geometry of the final compounds. The rotaxanes adopt one of two conformations: folded or extended. Rotaxanes are formed by simultaneous interactions of electron rich benzene rings with electron deficient six membered chelate rings and formation of hydrogen bonds between the secondary amino group and crown ether oxygens. Variable temperature 1H NMR studies of diamagnetic di-Ni-rotaxanes confirm that shuttling of the dibenzo-24-crown-8 moiety takes place along the p-xylylene linked axle, while folding of the axle in the m-xylylene linked rotaxane stabilizes the bent geometry of the whole compound and prevents shuttling of the crown ether moiety. In the case of rotaxanes containing the p-xylylene linkers, at elevated temperatures we observe a translocation of the crown ether unit between the metal coordinating macrocycles, which is well supported by electrochemical results for heteronuclear (Cu/Ni) systems but such translocation was not observed in the m-xylylene bridges. Electrochemical results suggest that an unfolding of the axle takes place upon oxidation of the metal centre in the di-Ni-rotaxane but not in heteronuclear m-xylylene linked rotaxanes.


 Please wait while we load your content...
Please wait while we load your content...